29 November 2022
By: Alison Crandell and Jax Karline
Introduction:
We are disappointed to announce that our time with RISEbio has come to an end with this blog post. The research we have been doing on the brain and behavior of green anole lizards is coming to an end, allowing us to move on to more concentrated work. During the breeding and nonbreeding season of male and female green anoles, changes in behavior and morphology can be observed. The cause for these differences in the brain is unknown, but may result from changes in gene expression. Based on the findings of scientific articles, we were able to make predictions for the roles that our chosen genes, EHMT2 and PLA2G1B, could play. Methyltransferase EHMT2 adds methyl tags to histones, repressing gene expression. EHMT2 inhibition has led to reduced anxiety in mice, and in anoles increased anxiety due to higher expression could explain the absence of mating behaviors in the nonbreeding season. Therefore, we predicted that EHMT2 would be upregulated in the nonbreeding season. Phospholipase A2 group IB (PLA2G1B) pathways are connected to metabolism, which the brain uses to secrete hormones through the pituitary gland to interact with sexual organs. Upregulation of PLA2G1B was expected in the breeding season due to the increased sexual displays. Having made these hypotheses nearly a year ago, we have conducted many experiments building up towards our final results. Now, we can finally see if our hypotheses held true, and present this information to others.
Body:
To quantify whether expression of our genes varied between sex or season for green anoles, we conducted a qPCR. This works by running through various PCR cycles, amplifying more and more cDNA each time. During each cycle the strands of DNA come apart, the primer attaches, and then a complementary strand is created that turns that single strand into a newly formed double stranded cDNA. SYBR green, which is added in the qPCR master mix, is a unique type of dye that fluoresces in the presence of double stranded DNA. So, at the end of each cycle, the qPCR machine can measure the amount of cDNA present based on the intensity of the fluorescent signals. These signals, and therefore the gene expression, are amplified during each cycle before eventually reaching a plateau. Samples that have a higher expression of a certain gene will reach this threshold cDNA concentration much faster. The actual amount for this threshold does not matter, although it needs to be above the noise and below the plateau. The final data collected from the qPCR was the amount of cDNA present in each well at the selected threshold value.
After verifying the validity of the qPCR through a melt curve and gel electrophoresis, we sent the data to Dr. Cohen to be decoded. This allowed us to know the sex and season of each sample instead of having them simply labeled by a number. After standardizing our data by dividing it by beta actin (a control gene that is equally expressed across sex and season), we were able to start with statistical analysis. First, we used a Grubb’s outlier test to determine if any samples were outliers. Then, we used a one-way ANOVA to assess the differences across all the groups. For EHMT2 there was a sex*season (p=.039) and sex (p<.001) difference. This means that there is higher EHMT2 expression in males, especially in the breeding season. This can be related back to the original hypothesis that higher EHMT2 expression is correlated with higher anxiety. Since males have the added pressure of finding mates during the breeding season, it makes sense that they could have higher anxiety from higher EHMT2 expression in the breeding season. For PLA2G1B, there was a seasonal difference (p<.001). This means that PLA2G1B mRNA expression differs seasonally in the hypothalamus of green anole lizards. The upregulation observed in the nonbreeding season may signal to slow down the metabolism, allowing lizards to store energy needed for the breeding season. After getting these results, we still had a time left to work in the lab, and picked up another gene, NPAS2, to work with. This gene was worked on by past RISEbio students who had already found a working primer but did not get a chance to finish their work with the gene. We were able to run a standard curve and qPCR for NPAS2, and after analyzing the results we saw that there was a sex*season difference.
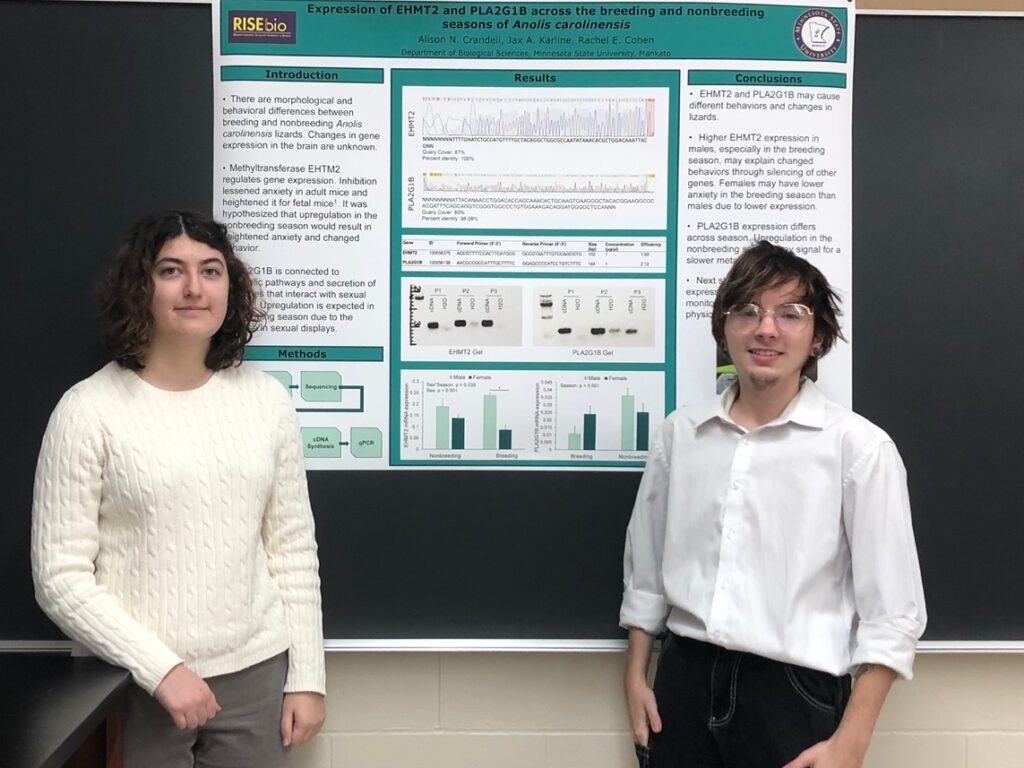
After we finished collecting data for our genes and interpreted what these findings could mean, we began working on creating posters to present this information to others. Scientists often attend conferences where many people gather to both present and view posters. This allows them to see novel work in their fields, view results that have yet to be published, to ask questions, and receive feedback from others. A poster session was held for us RISEbio students for this very purpose, simulating what we may encounter in our professional careers. This poster presentation was the culmination of all the work we had done thus far, allowing us to present our results and to see what other students had achieved. A lot of work went into creating our poster, such as making graphs that accurately represented our results, and tables that included important data, like efficiency values and primer sequences. Since we were still working on NPAS2 while we were making the poster, we chose not to include this gene on the poster. We were also very conscious of design and aesthetics while creating our poster. We wanted it to look professional, yet at the same be interesting and visually appealing enough to get people’s attention. As such, we chose a strict color scheme to stick to, and were careful in selecting the fonts we used. We were also careful with formatting, since any small mistakes or slight misalignments would have been glaringly obvious once the poster got printed out at its huge size. In addition to design, the presentation aspect is also important during a poster presentation. It is important to understand the audience, and how best to communicate with them. For this, we practiced giving short elevator speeches and fully going through the presentation.
Conclusion:
After a year and a half of dedicated work, significant results are finally starting to show. It has been a valuable experience working through the program here, and we are thankful to have been able to participate in it. We think about all those who have helped us along the way: Dr. Sharlin, Dr. Cohen, our TAs Quentin Phillips and Nick Shankey, our peer mentor Taylor Grossen, and the rest of RISEbio. The skills we have learned, felt, and experienced during this semester have been extremely valuable to us, and will be used both throughout our college and professional careers.


